Product Overview
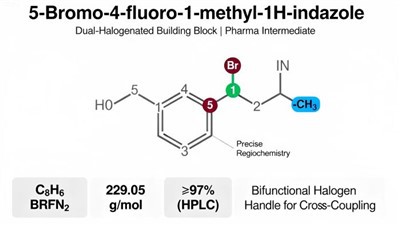
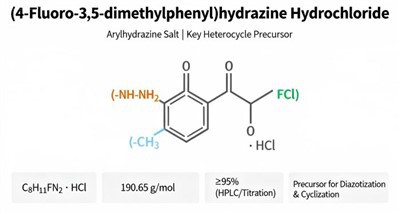
(S)-4-(4-Bromophenyl)-2,2-dimethyltetrahydro-2H-pyran is a premium, stereochemically defined building block designed for advanced pharmaceutical synthesis.
This chiral intermediate uniquely combines a reactive 4-bromophenyl group with a rigid, substituted tetrahydropyran (THP) ring in a specific (S)-configuration.
The bromo-aryl moiety serves as an excellent handle for pivotal cross-coupling reactions.
Such as Suzuki-Miyaura and Buchwald-Hartwig couplings, enabling rapid diversification of the aromatic core.

Simultaneously, the chiral THP ring provides a stable, three-dimensional oxygen-containing heterocycle that can significantly influence a molecule's solubility, metabolic profile, and target binding affinity.
Our production of (S)-4-(4-Bromophenyl)-2,2-dimethyltetrahydro-2H-pyran is controlled to deliver high chemical and enantiomeric purity, ensuring consistent performance in demanding asymmetric synthesis. This compound is an invaluable tool for medicinal chemists seeking to introduce both a versatile coupling site and a chiral, conformationally constrained fragment in a single step. It finds critical application in the research and development of targeted therapies, particularly where the spatial orientation of the aryl group is crucial for biological activity.
About us

why choose us
Expert Consultation & Education
Access to our dedicated medical affairs team for scientific data and clinical studies.
Marketing & Sales Support
Clear documentation on product indications and contraindications and Flexible ordering options and transparent pricing quotes.
Efficient Order Processing & Logistics
User-friendly online ordering portal with real-time tracking.
Warranty & Replacement Program
Product Satisfaction Guarantee. We stand behind our products.
Product packaging
Thank you for your trust,we are committed to ensuring every order meets your expectations.We will process your order on the same day. All products undergo meticulous inspection and are packaged securely to ensure they arrive in perfect condition.




FAQ
Q: What are the key synthetic advantages of this intermediate?
+
-
A: This molecule offers two key advantages in one structure: 1) A bromine atom for reliable metal-catalyzed cross-coupling to introduce complex fragments, and 2) A pre-formed chiral tetrahydropyran ring that acts as a rigid, bioactive scaffold. Using (S)-4-(4-Bromophenyl)-2,2-dimethyltetrahydro-2H-pyran can streamline synthesis by avoiding separate steps to create chirality and install the heterocycle.
Q: How do you ensure the stereochemical integrity (S-configuration) of the product?
+
-
A: We employ stereoselective synthesis or chiral resolution techniques followed by rigorous analytical control. The enantiomeric purity of each batch of (S)-4-(4-Bromophenyl)-2,2-dimethyltetrahydro-2H-pyran is verified using chiral HPLC or SFC, with a detailed Certificate of Analysis provided to guarantee the specified configuration and purity for your asymmetric synthesis needs.
Q: Can you provide related derivatives or custom analogs of this chiral building block?
+
-
A: Yes. Our expertise extends to custom synthesis. We can develop a range of derivatives based on this core structure, including compounds with different aryl halides (e.g., iodophenyl) or further functionalization of the THP ring, tailored to support your specific drug discovery programs.
We are committed to supplying high-value chiral intermediates for innovative drug development. Contact us for specifications, pricing, and sample availability of (S)-4-(4-Bromophenyl)-2,2-dimethyltetrahydro-2H-pyran.
Q: Can you do our design?
+
-
A: Of course, your customized design (OEM) is available.
Q: Can you make our design for the package?
+
-
A: Yes, please send us the design you need ,we will value the price and make the exact same package basing on your design. MOQ 1000pcs.
Hot Tags: (s)-4-(4-bromophenyl)-2,2-dimethyltetrahydro-2h-pyran, China (s)-4-(4-bromophenyl)-2,2-dimethyltetrahydro-2h-pyran manufacturers, suppliers, R 4 methyl 1 3 2 dioxathiolane 2 2 dioxide, S 3 Aminobutanenitrile Hydrochloride, S 4 4 bromophenyl 2 2 dimethyltetrahydro 2H pyran, 1 4 dibromo 2 fluorobenzene, 5-Bromo-2-fluoro-m-xylene, 5-Bromo-4-fluoro-1-methyl-1H-indazole